Understanding Curie and Becquerel: Essential Radiation Activity Units
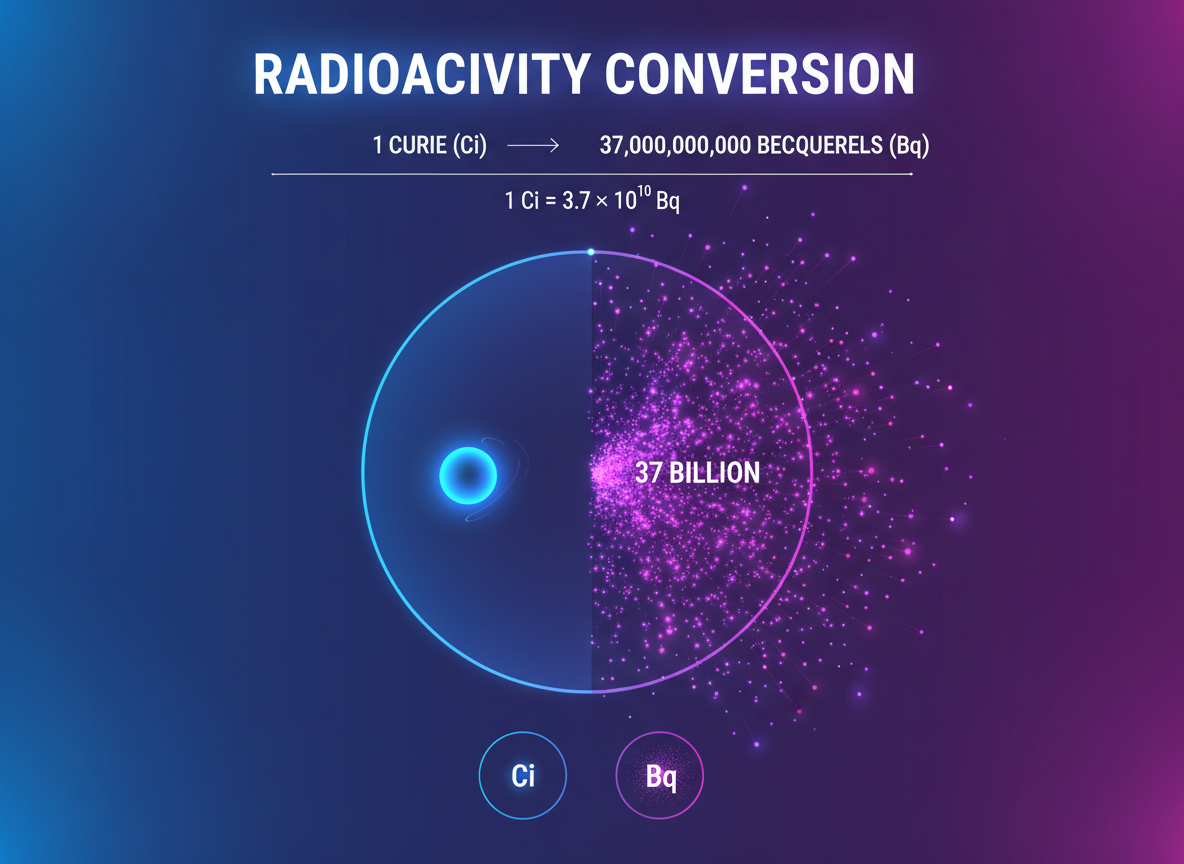
When you’re working with radioactive materials—whether in a hospital, research lab, or nuclear facility—you’ll need to convert between 1 Curie to Bq (Becquerel) for accurate measurements. Here’s the exact answer: 1 Curie equals 37,000,000,000 Bq, or 37 billion Becquerels.
This conversion matters tremendously. Medical professionals use it when calibrating radiation therapy equipment, nuclear engineers rely on it for safety protocols, and researchers need it for experimental documentation. The Curie, named after Marie and Pierre Curie, originated from early radioactivity research and represented the activity of 1 gram of radium-226. The Becquerel, named after Henri Becquerel who discovered radioactivity, became the SI standard unit in 1975.
Today, most scientific literature uses Becquerels, while Curies remain common in medical applications and older nuclear facilities. Understanding both units—and converting between them—isn’t just academic. It’s essential for safety, compliance, and professional communication. Just as engineers need precise micro-scale conversions, radiation professionals need flawless activity measurements.
Quick Answer: 1 Curie to Bq Conversion
Let’s cut straight to what you’re looking for:
1 Curie (Ci) = 37,000,000,000 Becquerels (Bq)
You can also write this as 3.7 × 10¹⁰ Bq in scientific notation, which makes handling these large numbers much easier.
The conversion formula is straightforward:
Becquerels = Curies × 37,000,000,000
Or in scientific notation: Bq = Ci × 3.7 × 10¹⁰
Here’s a quick reference table for common conversions:
| Curies (Ci) | Becquerels (Bq) | Scientific Notation |
|————-|—————–|———————|
| 0.001 | 37,000,000 | 3.7 × 10⁷ Bq |
| 0.01 | 370,000,000 | 3.7 × 10⁸ Bq |
| 0.1 | 3,700,000,000 | 3.7 × 10⁹ Bq |
| 1 | 37,000,000,000 | 3.7 × 10¹⁰ Bq |
| 10 | 370,000,000,000 | 3.7 × 10¹¹ Bq |
That’s a huge difference between units, right? The Curie represents significantly more radioactive decay events than a single Becquerel. If you’re working with other technical conversions—like converting 150 grain to mg for ballistics calculations—you’ll notice that radiation units often involve similarly large scaling factors.
What Is a Curie (Ci)? History and Definition
The curie (Ci) is a non-SI unit of radioactivity that honors Marie and Pierre Curie, the pioneering scientists who revolutionized our understanding of radiation. It’s a measurement that’s been around since 1910, making it quite the veteran in the world of nuclear science.
Originally, the curie was defined by the radioactive activity of 1 gram of radium-226, the element that the Curies spent years isolating and studying. This definition made sense at the time since radium-226 was the primary reference standard for radioactivity.
Today, we define one curie more precisely: it’s exactly 3.7 × 10¹⁰ (37 billion) radioactive decays per second. That’s an enormous number. To put it in perspective, each curie represents 37 billion atomic nuclei disintegrating every single second. This specific value was chosen because it closely matched the activity of that original gram of radium-226.
The curie measures radioactive decay rate, which tells us how “active” a radioactive source is. Whether you’re working with uranium, cobalt-60, or any other radioactive material, the curie quantifies how many atoms are breaking down each second.
Despite the scientific community’s shift toward SI units (where the becquerel is standard), the curie remains popular in several fields. Nuclear medicine, radiation therapy, and certain industrial applications in the United States still frequently use curies. It’s similar to how measurement standards in other fields maintain legacy units—just as we discussed in our guide on liter to kiloliter conversions, sometimes the older unit just sticks around.
What Is a Becquerel (Bq)? The SI Standard Unit

The becquerel (Bq) is the modern SI unit of radioactivity, named after French physicist Henri Becquerel, who discovered radioactivity in 1896. Unlike the curie’s historical complexity, the becquerel uses an elegantly simple definition: 1 Bq equals exactly 1 radioactive decay per second. That’s it—no arbitrary multipliers or complicated origins.
This straightforward approach makes the becquerel perfect for scientific communication across borders. When researchers in Tokyo, Toronto, or Tel Aviv publish findings, they’re all speaking the same measurement language. The SI standardization removes ambiguity and streamlines collaboration—similar to how other SI unit conversions provide universal clarity in measurements.
Of course, measuring one decay per second isn’t always practical. That’s where convenient multiples come in:
- kBq (kilobecquerel) = 1,000 Bq
- MBq (megabecquerel) = 1,000,000 Bq
- GBq (gigabecquerel) = 1,000,000,000 Bq
- TBq (terabecquerel) = 1,000,000,000,000 Bq
Today’s scientific literature overwhelmingly favors the becquerel. Medical imaging reports, environmental monitoring data, and research papers published after the 1970s typically use Bq units. While the curie still appears in older texts and some specialized fields, the becquerel represents where radiation measurement is headed—and where it’s been for most working scientists since global adoption began decades ago.
Conversion Formula and Step-by-Step Calculation
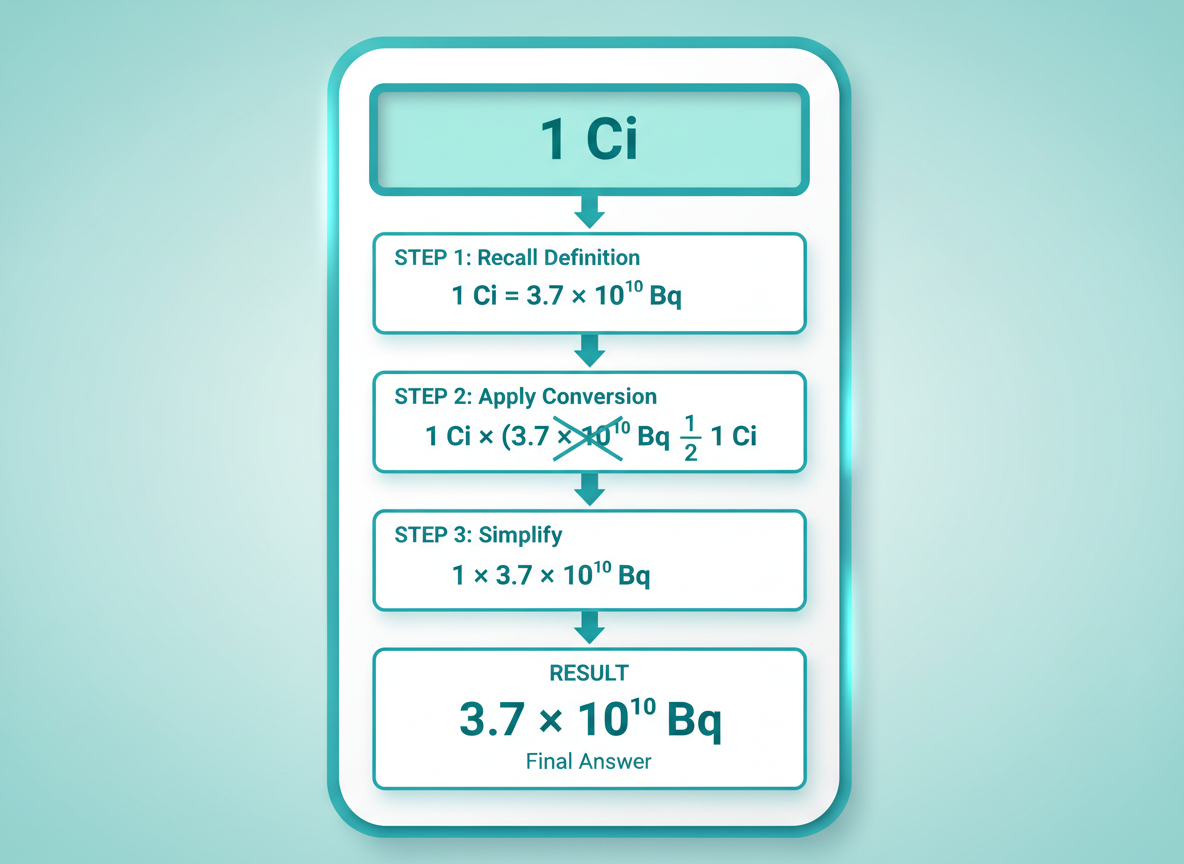
The math here isn’t complicated, but those big numbers can look intimidating at first glance. Here’s the formula you’ll need:
Bq = Ci × 3.7 × 10¹⁰
To go the other direction:
Ci = Bq ÷ 3.7 × 10¹⁰
Let’s walk through converting 1 Curie to Becquerels step by step.
Example: Converting 1 Curie
1. Start with your value: 1 Ci
2. Multiply by 3.7 × 10¹⁰
3. Result: 37,000,000,000 Bq (or 3.7 × 10¹⁰ Bq)
That’s 37 billion Becquerels! This massive number shows why the Curie remains popular in certain fields—it’s simply more convenient for high-activity sources.
Working with Scientific Notation
When you’re dealing with numbers this large, scientific notation is your friend. Keep your calculator in scientific mode, and you’ll avoid errors from missing zeros. For instance, 3.7 × 10¹⁰ is much cleaner than writing out 37,000,000,000.
Precision Matters
For scientific work, maintain at least three significant figures throughout your calculation. If you’re converting 2.5 Ci, your answer should be 9.25 × 10¹⁰ Bq, not just “about 90 billion.”
However, context matters. In regulatory documentation, you’ll often need full precision. For general understanding or educational purposes, rounding to two or three significant figures usually works fine.
Much like converting 1.1 Bar to PSI, the key is understanding your formula and maintaining appropriate precision for your specific application.
Real-World Applications and Measurement Examples
Radiation activity measurements touch nearly every aspect of modern life, from hospitals to power plants. Understanding typical activity levels helps put the curie-to-becquerel conversion into perspective.
Medical Applications
Hospitals routinely work with radioactive sources for both diagnosis and treatment. A typical cobalt-60 radiotherapy source might contain 200-400 Ci (7.4-14.8 TBq), delivering precise radiation doses to cancer cells. Diagnostic imaging uses much smaller amounts—technetium-99m for bone scans typically involves 20-30 mCi (740-1,110 MBq) per patient dose. Iodine-131 thyroid treatments fall somewhere between, usually around 5-200 mCi (185-7,400 MBq) depending on the condition.
Nuclear Power and Research
Nuclear fuel rods present an entirely different scale. A single spent fuel assembly can exceed millions of curies of activity, making becquerels the more practical unit at such levels. Reactor monitoring systems track everything from neutron flux to cooling water contamination, often measuring in kilobecquerels (kBq) or megabecquerels (MBq).
Research laboratories handle more manageable quantities. Carbon-14 dating samples might contain just 0.001-0.01 µCi (37-370 Bq). Radiotracer experiments commonly use 1-10 µCi (37-370 kBq) to track chemical pathways in biological systems.
Industrial and Environmental Uses
Industrial radiography for pipeline inspections typically employs iridium-192 sources at 10-100 Ci (370-3,700 GBq). Food and medical equipment sterilization facilities use massive cobalt-60 arrays reaching thousands of curies.
Environmental monitoring reveals the world around us. Background radiation in most locations measures about 0.1-0.2 µSv/h, translating to roughly 1-10 Bq per kilogram of soil. Contamination assessments after nuclear incidents require precise measurements—similar to other practical measurement applications—where understanding both units becomes essential for safety protocols.
These diverse applications show why converting between curies and becquerels matters across so many fields.
Curie vs Becquerel: When to Use Each Unit
The scientific community overwhelmingly prefers Becquerels. Most peer-reviewed journals and international research publications require SI units, making Bq the standard for submitting papers. If you’re writing for Nature, Science, or similar outlets, stick with Becquerels.
However, the United States nuclear industry and some medical facilities still rely heavily on Curies. You’ll find mCi (millicuries) commonly used in American hospital radiation therapy departments and nuclear medicine labs. This isn’t stubbornness—it’s practical. Decades of protocols, safety training, and documentation use Curies, and converting everything would be expensive and potentially confusing during the transition.
Regulatory requirements vary by region. The International Atomic Energy Agency (IAEA) mandates Becquerels, while the U.S. Nuclear Regulatory Commission accepts both units but traditionally leans toward Curies in legacy documents. European regulations exclusively use Becquerels.
For your audience, choose wisely. Presenting to an international conference? Use Bq. Working with American radiation safety officers? They’ll probably appreciate Ci alongside Bq conversions. Teaching students? Show both units but emphasize Becquerels as the modern standard.
Much like understanding unit system differences between inches and millimeters, knowing when to use Curie versus Becquerel depends on your context and audience. When in doubt, provide both—it’s better to be clear than technically “correct” by one system alone.
Quick Reference Conversion Table
Here’s your go-to table for converting Curie values to Becquerels, covering everything from tiny medical doses to industrial-strength sources:
| Curie (Ci) | Becquerel (Bq) | Practical Application |
|————|—————-|———————-|
| 0.000001 Ci (1 µCi) | 3.7 × 10⁴ Bq (37 kBq) | Medical tracer studies |
| 0.001 Ci (1 mCi) | 3.7 × 10⁷ Bq (37 MBq) | Diagnostic thyroid scans |
| 0.01 Ci (10 mCi) | 3.7 × 10⁸ Bq (370 MBq) | Cancer treatment seeds |
| 0.1 Ci | 3.7 × 10⁹ Bq (3.7 GBq) | Industrial gauges |
| 1 Ci | 3.7 × 10¹⁰ Bq (37 GBq) | Calibration sources |
| 10 Ci | 3.7 × 10¹¹ Bq (370 GBq) | Industrial radiography |
| 100 Ci | 3.7 × 10¹² Bq (3.7 TBq) | Food irradiation |
| 1,000 Ci | 3.7 × 10¹³ Bq (37 TBq) | Research reactors |
Bookmark this table for instant conversions—it’s as handy as our 1 1/3 Cup to Liters converter for kitchen measurements. Need different values? Unit Genie handles any conversion you throw at it.
Understanding Radioactivity and Decay Rates
Radioactive decay isn’t just a number—it’s nature’s way of unstable atoms seeking stability. When we measure activity in Bq or Curies, we’re counting how many atoms transform per second, releasing radiation in the process. Think of it like popcorn kernels popping: activity tells you the popping rate, not how much popcorn you started with.
Here’s where half-life enters the picture. A substance with a short half-life has frantic atomic decay (high activity), while longer half-lives mean slower, steadier transformations. Iodine-131, used in thyroid treatments, has an 8-day half-life and relatively high activity per gram. Compare that to uranium-238’s 4.5-billion-year half-life—its activity per gram is remarkably low.
This brings us to specific activity: the relationship between how much material you have and how radioactive it is. Two samples might show identical activity readings of 37 GBq (1 Ci), but one could be a tiny speck while the other fills a beaker. The measurement precision required here mirrors other laboratory work, similar to how 0.1 mL to microliters conversions demand accuracy in medical settings.
Now, let’s clear up a common confusion: activity, dose, and exposure aren’t interchangeable. Activity measures the source’s strength. Dose (measured in Sieverts or rems) indicates how much radiation energy your body absorbs. Exposure describes your radiation environment. You could stand near a high-activity source and receive minimal dose if properly shielded—distance and barriers matter tremendously.
When interpreting measurements, context is everything. A 3.7 MBq (100 µCi) smoke detector is perfectly safe. That same activity from an unshielded medical isotope in your pocket? That’s a problem.
Safety Context and Practical Considerations
Here’s something vital to understand: measuring radioactivity in becquerels or curies tells you how many atoms are decaying, but it doesn’t tell you how much radiation you’re actually absorbing. That’s a completely different measurement called dose, expressed in sieverts or rems.
Think of it this way—a 10 Bq source could be relatively harmless if properly contained, while even low-activity materials can pose risks if ingested or inhaled. Context matters enormously.
Generally speaking, natural background radiation exposes you to about 3-4 millisieverts per year. Consumer products like smoke detectors contain sources around 37 kBq (1 microcurie). Medical procedures might use sources from hundreds of megabecquerels to several gigabecquerels. Industrial applications can reach terabecquerel levels—that’s where serious shielding becomes non-negotiable.
The NRC and IAEA establish strict regulatory limits for occupational exposure (typically 50 millisieverts per year) and public exposure (1 millisievert per year). These organizations also define what constitutes exempt quantities, which vary by isotope.
Just like you’d want precision when converting measurements—say, using a grain to mg calculator for pharmaceutical applications—you need expert guidance for radiation safety. Never handle radioactive materials without proper training, shielding, and monitoring equipment. If you’re working with radiation sources professionally, always consult qualified radiation safety officers and follow your institution’s protocols. Your health depends on it.
Frequently Asked Questions (FAQ)
How many Becquerels are in 1 Curie?
1 Curie equals exactly 37,000,000,000 Becquerels (3.7 × 10¹⁰ Bq). The formula is straightforward: multiply Curies by 37 billion to get Becquerels. Going the other direction, divide Becquerels by 37 billion to get Curies.
Why is the conversion factor 37 billion?
The number comes from Marie Curie’s original research with radium. Scientists defined 1 Curie as the activity of exactly 1 gram of radium-226, which undergoes approximately 37 billion disintegrations per second. When the Becquerel became the SI standard unit (measuring 1 disintegration per second), this historical measurement created the conversion factor we use today.
Which unit is more commonly used today?
The Becquerel dominates in countries using the International System of Units, including most of Europe, Asia, and scientific publications worldwide. However, the Curie still appears in older literature, medical facilities in the United States, and legacy nuclear documentation. You’ll often see both units listed side-by-side for clarity.
How do I convert millicuries to Becquerels?
One millicurie (mCi) equals 37,000,000 Bq or 37 megabecquerels (MBq). Multiply your millicuries by 37 million. For example, 5 mCi × 37,000,000 = 185,000,000 Bq (185 MBq). This conversion comes up frequently in medical imaging procedures.
What’s the difference between Curie and radiation dose units?
Curies and Becquerels measure activity—how many atoms decay per second in a radioactive source. They don’t measure exposure or dose. Units like Sieverts, rems, and Grays measure the radiation’s effect on tissue. A highly active source (measured in Curies) might pose minimal risk if properly shielded, while lower activity could be dangerous depending on radiation type and distance.
Is 1 Curie dangerous?
Context matters tremendously. 1 Curie represents extremely high activity—37 billion decays every second. Whether it’s dangerous depends on the radiation type (alpha, beta, gamma), exposure duration, distance, and shielding. This level typically requires professional handling, specialized containment, and safety protocols. Never assume based solely on activity numbers.
Can I use this conversion for all radioactive materials?
Yes! Activity measurements apply universally across all radioactive isotopes. Whether you’re working with carbon-14, cesium-137, or uranium-235, the Curie-to-Becquerel conversion remains constant at 3.7 × 10¹⁰.
What instruments measure radioactivity in these units?
Geiger-Müller counters, scintillation detectors, and ionization chambers all measure radioactivity. Modern devices typically display readings in Becquerels, though many offer switchable units. Professional equipment can measure across wide ranges, from background radiation (around 0.1 μSv/h) to industrial sources. Find more conversion calculators and guides at Unit Genie’s blog for scientific, engineering, and everyday measurements.



